It starts as a small red bump on your leg or arm. You might think it’s just a spider bite or an ingrown hair. But days later, it turns into a painful, swollen abscess that refuses to heal. This is the classic presentation of MRSA, which stands for Methicillin-Resistant Staphylococcus aureus. It is a strain of bacteria that has evolved to resist many common antibiotics. For decades, doctors treated MRSA as a problem confined to hospitals. Today, that line is blurred. You can catch it at the gym, in a locker room, or even at home. Understanding where MRSA comes from and how it spreads is no longer just medical trivia-it is essential for protecting your health.
The distinction between community-associated MRSA (CA-MRSA) and healthcare-associated MRSA (HA-MRSA) used to be clear-cut. Now, it is a moving target. The Centers for Disease Control and Prevention (CDC) defines CA-MRSA as an infection diagnosed in someone who has not been hospitalized, had surgery, or lived in a nursing home in the past year. Yet, recent data shows that nearly 1.3% of people living in the community are colonized with MRSA. This means they carry the bacteria on their skin or in their nose without showing symptoms, acting as silent carriers who can spread it to others. The old rules don't work anymore because the bacteria have adapted to thrive outside the sterile environment of a hospital.
Genetic Differences Between CA-MRSA and HA-MRSA
To understand why these infections behave differently, you have to look under the microscope at their genetic makeup. Both strains belong to the family of Staphylococcus aureus, but they carry different genetic toolkits. The key difference lies in a piece of DNA called the staphylococcal cassette chromosome mec, or SCCmec. This genetic element gives the bacteria its resistance to methicillin and related drugs like penicillin and amoxicillin.
Community-associated MRSA typically carries smaller versions of this genetic cassette, known as types IV and V. Because these elements are smaller, they leave more room for other genes-specifically, those that code for toxins. One of the most dangerous is Panton-Valentine leukocidin (PVL). This toxin destroys white blood cells, allowing the bacteria to cause rapid tissue damage. That is why CA-MRSA often presents as aggressive skin infections or necrotizing pneumonia. In contrast, hospital-associated MRSA usually carries larger SCCmec types (I, II, or III). These larger cassettes come with a heavy baggage of resistance genes, making HA-MRSA resistant to a wider array of antibiotics, including erythromycin, clindamycin, and fluoroquinolones.
In the United States, one specific clone dominates the community landscape: USA300. This strain accounts for roughly 70% of CA-MRSA cases. It is highly transmissible and virulent, spreading easily through direct skin-to-skin contact. Meanwhile, in hospitals, strains like ST239 and the emerging ST59 are more common, particularly in regions like China and Europe. These differences matter because they dictate how doctors treat you. A treatment that works for a community strain might fail against a hospital strain due to multi-drug resistance.
| Feature | CA-MRSA | HA-MRSA |
|---|---|---|
| Primary Setting | Gyms, schools, households | Hospitals, nursing homes |
| SCCmec Type | Types IV and V (smaller) | Types I, II, III (larger) |
| Key Toxin | Panton-Valentine leukocidin (PVL) | Rarely produces PVL |
| Antibiotic Resistance | Often susceptible to clindamycin and TMP-SMX | Frequently multi-drug resistant |
| Typical Patient | Healthy individuals, athletes | Immunocompromised, post-surgery |
How MRSA Spreads in the Community
You do not need a weakened immune system to get MRSA. In fact, CA-MRSA thrives among healthy people. The primary mode of transmission is direct skin-to-skin contact. Think about activities that involve close physical proximity: wrestling, rugby, football, or sharing towels and razors in a dormitory. Any break in the skin-a cut, scrape, or acne lesion-provides an entry point for the bacteria.
Certain environments act as super-spreader zones. Studies have shown that people living in military barracks face a 12.3 times higher risk of colonization compared to the general population. Homeless shelters see an 8.7 times higher risk, while prisons present a staggering 14.9 times higher risk. Crowding, poor sanitation, and frequent skin trauma create a perfect storm for transmission. Injecting drug users are another high-risk group, not just because of shared needles, but because frequent punctures compromise the skin barrier, allowing MRSA to enter the bloodstream.
The scary part is that many people are asymptomatic carriers. They harbor the bacteria in their anterior nares (the front of the nose) or on their groin area without knowing it. When they scratch or touch their skin, they transfer the bacteria to surfaces or other people. This is why public health experts emphasize hygiene. Washing hands regularly, keeping cuts covered with clean bandages, and avoiding sharing personal items like sports equipment or clothing can significantly reduce your risk.
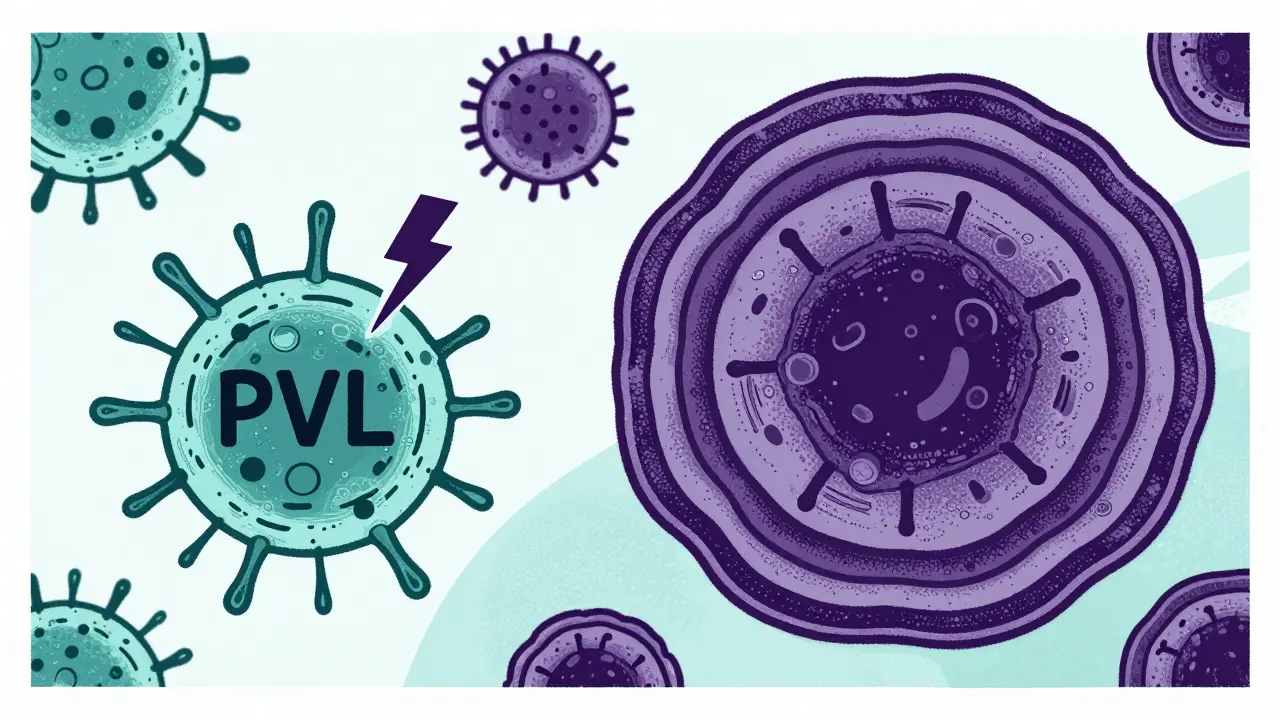
The Blurring Line: Hospital-Community Transmission
For years, infection control protocols assumed that MRSA stayed in the hospital. If you got it there, it was HA-MRSA. If you got it outside, it was CA-MRSA. Recent research shatters this assumption. A study conducted in Alberta, Canada, found that 27.6% of hospital-onset MRSA infections were caused by CA-MRSA strains. Conversely, 27.5% of community-associated infections were caused by HA-MRSA strains. This bidirectional flow means that patients bring community strains into hospitals, and discharged patients take hospital strains back into the community.
Why does this happen? The average hospital stay is now quite short, often just four to five days. However, it can take several hundred days for a person to naturally clear MRSA from their body after discharge. During this window, they remain contagious. Furthermore, healthcare workers themselves can act as vectors, carrying strains between settings. Mathematical models suggest that this coexistence is fragile but stable. Without intervention, highly transmissible community strains like USA300 could eventually dominate hospital wards, displacing traditional hospital strains. This shift forces hospitals to rethink their surveillance strategies, focusing less on isolating "hospital" bugs and more on monitoring all incoming patients for colonization.

Treatment Strategies and Antibiotic Choices
Treating MRSA requires precision. Using the wrong antibiotic can lead to treatment failure and worsen the infection. For mild skin infections caused by CA-MRSA, incision and drainage (I&D) is often sufficient. The pus needs to be removed physically. If antibiotics are necessary, doctors typically choose oral medications that target the specific resistance profile of CA-MRSA. Trimethoprim-sulfamethoxazole (TMP-SMX) and clindamycin are common first-line choices. Approximately 96% of CA-MRSA isolates remain susceptible to clindamycin, making it a reliable option in many regions. Tetracyclines, such as doxycycline, are also effective, with susceptibility rates around 89%.
However, if the infection is severe, involves the bloodstream (bacteremia), or originates from a hospital setting, the stakes are much higher. HA-MRSA is notoriously resistant to multiple drug classes. Intravenous antibiotics like vancomycin or daptomycin become necessary. Vancomycin has been the gold standard for decades, but resistance is creeping up, prompting researchers to monitor minimum inhibitory concentrations (MICs) closely. Daptomycin offers an alternative mechanism of action, disrupting the bacterial cell membrane. For complicated pneumonia, linezolid may be preferred because it penetrates lung tissue effectively.
A major challenge is the emergence of hybrid strains. These are CA-MRSA clones that have acquired additional resistance genes, giving them the virulence of community strains and the resilience of hospital strains. When treating a patient, clinicians must rely on culture and sensitivity testing rather than guessing. Empirical therapy-starting treatment before test results return-must account for local resistance patterns. In areas where USA300 is prevalent, assuming susceptibility to beta-lactams is a dangerous error.
Prevention and Future Outlook
Preventing MRSA requires a two-pronged approach: individual hygiene and systemic surveillance. On a personal level, the basics still apply. Shower after exercising, especially in communal gyms. Do not share personal items like towels, barbershop tools, or athletic gear. Keep any wounds clean and covered until healed. If you have a boil or abscess, seek medical attention early; do not try to pop it yourself, as this can push bacteria deeper into the tissue.
On a broader scale, the medical community is shifting toward integrated surveillance. Instead of treating hospital and community reservoirs as separate entities, public health agencies are tracking MRSA across the entire continuum. Decolonization protocols, which involve using antiseptic washes like chlorhexidine and nasal ointments like mupirocin, are being explored for high-risk patients before surgeries. However, overuse of these agents can drive resistance, so they must be used judiciously.
The future of MRSA management depends on reducing unnecessary antibiotic use. Every time we prescribe an antibiotic for a viral infection, we select for resistant bacteria. By preserving the effectiveness of our current arsenal, we buy time for new drugs and therapies to develop. Until then, awareness remains our best defense. Recognizing the signs of a staph infection early and understanding that MRSA is no longer just a "hospital bug" can save lives.
What are the first signs of a MRSA infection?
Early signs often resemble a spider bite or a pimple that grows larger, becomes red, swollen, and painful. As it progresses, it may turn into an abscess filled with pus. Systemic symptoms like fever, chills, and fatigue may indicate the infection has spread beyond the skin.
Can you get MRSA from a toilet seat?
It is unlikely. MRSA primarily spreads through direct skin-to-skin contact or contact with contaminated items like towels, razors, or sports equipment. While the bacteria can survive on surfaces, casual contact with a toilet seat is not a common transmission route.
Is MRSA curable?
Yes, MRSA is treatable. Mild skin infections often respond to incision and drainage combined with appropriate oral antibiotics. Severe infections require intravenous antibiotics like vancomycin. Early diagnosis and proper treatment are critical to preventing complications.
How long does it take to clear MRSA from the body?
After successful treatment of an active infection, the bacteria may persist on the skin or in the nose for weeks or months. Natural clearance can take several hundred days. Recolonization is possible, especially if exposed to infected individuals or contaminated environments.
What is the difference between CA-MRSA and HA-MRSA?
CA-MRSA affects healthy people in the community and is often more virulent due to toxins like PVL. HA-MRSA affects patients in healthcare settings and is typically more resistant to multiple antibiotics. Genetic differences in SCCmec types distinguish the two strains.